Strategies to improve dentin bonding
Key Highlights
- Proper surface preparation, including instrument choice and smear layer management, is essential for effective dentin bonding, especially with self-etch systems.
- Two-bottle bonding systems allow better control over primer and adhesive application, with thorough solvent evaporation being critical for bond strength.
- Active agitation during adhesive application enhances infiltration and bond quality, while adequate light curing ensures complete polymerization of the adhesive layer.
- Controlling moisture content post-etching and avoiding over-etching prevents collagen collapse and preserves hybrid layer integrity for durable bonds.
- Applying MMP inhibitors like chlorhexidine can inhibit enzymatic degradation, improving the long-term stability of the hybrid layer.
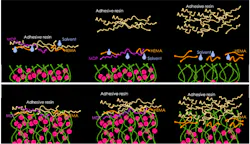
In many medium- and large-sized direct restorations, the surface area of exposed dentin may exceed that of enamel. To adequately resist the stresses generated by composite polymerization shrinkage and to transfer occlusal and thermal forces throughout the restoration, a durable dentin bond is essential. Dentin bonding is achieved through the formation of both a hybrid layer and resin tags. The hybrid layer forms when the outer layer of dentin is demineralized, exposing a network of collagen fibrils that are subsequently infiltrated with adhesive resin and polymerized. Resin tags are created when adhesive penetrates into the dentinal tubules.1 Bonding to dentin is considerably more technique sensitive than enamel because it is a hydrated, organic, and histologically variable structure that requires precise control of moisture, infiltration, and solvent evaporation to achieve stable hybridization and avoid incomplete resin penetration and long-term degradation.2 This article reviews several critical steps involved in optimizing dentin bonding.
Surface preparation prior to bonding
The instrument used to prepare dentin influences bond strength due to its effect on the smear layer. The smear layer consists of debris and particles of ground dentin that remain on the tooth surface following preparation with rotary instruments. Smear layer thickness is greatest with coarse diamond burs, followed by fine diamonds, and is least with carbide burs.3 The clinical significance of the smear layer depends on the bonding strategy employed. In a total-etch technique, phosphoric acid removes the smear layer entirely, resulting in similar bonding outcomes regardless of the instrument used.4 In contrast, with self-etch systems, the acidic adhesive must penetrate and solubilize the smear layer. Thick smear layers may inhibit adequate penetration, potentially reducing bond strength.4 Therefore, when using self-etch adhesives, final preparation with carbide burs or fine diamond instruments is preferred.
Air abrasion (i.e., sandblasting) is another method to prepare the surface of dentin for bonding. This method may be effective for cleaning any contamination from the surface of the dentin prior to bonding; however, laboratory testing indicates that air abrasion does not affect dentin bond strength negatively or positively.5 Recent research suggests that air abrasion used with concomitant irrigation improves bond strength.6
Single vs. two-bottle bonding agents
In a two-bottle bonding system, the first bottle contains the primer, which typically consists of hydrophilic resin monomers (such as 2-hydroxyethyl methacrylate, HEMA) dissolved in volatile solvents such as ethanol, acetone, or water. The hydrophilic monomers allow penetration into the wet environment of the exposed collagen network to form the initial hybridized zone. The solvents facilitate transport of the monomers into demineralized dentin by reducing the primer viscosity and acting as a hydrophilic carrier phase that is miscible with the water-rich collagen matrix, allowing monomers to diffuse into the interfibrillar collagen spaces; they also displace residual moisture and promote evaporation because of their high volatility.1 The second bottle contains the more hydrophobic adhesive resin, composed primarily of dimethacrylate monomers (such as bisphenol-A glycidyl dimethacrylate, Bis-GMA), viscosity regulators (triethylene glycol dimethacrylate, TEGDMA), photoinitiators, and, in some formulations, fillers.1 Upon light curing, this adhesive polymerizes to form a resin matrix that encapsulates collagen fibrils and copolymerizes with the restorative resin. In these systems, thorough solvent evaporation is critical during the priming step, whereas the adhesive layer primarily requires prevention of pooling and adequate light polymerization.
Single-bottle systems combine the primer and adhesive into one solution to simplify clinical procedures and reduce chairside time. However, because hydrophilic and hydrophobic components coexist in a single formulation, these systems may demonstrate increased water sorption over time.7 Additionally, solvent evaporation may be more challenging due to the mixed composition.
Total-etch vs. self-etch
In total-etch (etch-and-rinse) techniques, phosphoric acid is applied to dentin to remove hydroxyapatite crystals and expose a collagen matrix. In self-etch systems (or selective-etch techniques, in which enamel is etched but dentin is not), the bonding agent (the primer in a two-bottle system) contains acidic functional monomers (i.e., MDP, 10-methacryloyloxydecyl dihydrogen phosphate). These acidic monomers condition and prime dentin simultaneously, demineralizing the underlying dentin, dissolving the smear layer, and incorporating it into the hybrid layer.8 Universal adhesives are mildly acidic self-etch bonding agents that can also be used following phosphoric acid etching.
Microscopic evaluation demonstrates that total-etch systems typically produce a hybrid layer approximately 3-4 µm thick, whereas self-etch adhesives generally produce a thinner hybrid layer of approximately 1 µm.1 Despite this shallower penetration, self-etch adhesives may achieve comparable dentin bond strengths9 due to chemical interaction between functional monomers (e.g., MDP) and residual hydroxyapatite-bound calcium. The choice of etching technique may be affected by the type of dentin. For example, a clinical trial of noncarious cervical lesions indicated improved retention when a universal adhesive was applied in total-etch technique rather than self-etch or selective-etch technique, possibly a result of bonding to sclerotic dentin.10
Length of time for etching
With total-etch techniques, dentin should not be etched beyond the manufacturer’s recommended time (typically 15 seconds). Overetching may create a zone of demineralized collagen that exceeds the depth of adhesive infiltration, leaving unsupported collagen fibrils beneath the hybrid layer.11 These areas are susceptible to hydrolytic and enzymatic degradation, compromising long-term bond durability.
Moisture content after etching
Following etching in total-etch techniques, convention has been for dentin to remain visibly moist. This moisture prevents collapse of the exposed collagen network (desiccation) and allows adhesive monomers to infiltrate effectively. Many contemporary adhesives, including universal adhesives, contain water/ethanol solvent rather than acetone solvents and can rehydrate partially collapsed collagen, making precise moisture control somewhat less critical.12,13 Self-etch systems do not require premoistened dentin, and laboratory research suggest that application to gently air-dried dentin is acceptable.12
Active agitation of the bonding agent
Active agitation during adhesive application enhances infiltration between collagen fibrils and improves adaptation to the dentin surface. In self-etch systems, agitation also promotes dissolution and incorporation of the smear layer. Studies consistently demonstrate higher bond strengths when adhesives are actively scrubbed onto the dentin surface rather than passively applied.14,15
Air evaporation of the bonding agent
Air thinning is essential to evaporate residual solvent from the adhesive layer prior to polymerization. Incomplete solvent removal can lead to void formation and reduced bond strength.16 In two-bottle systems, solvent is typically present only in the primer; therefore, the priming step requires thorough air drying to ensure solvent evaporation. The adhesive layer primarily requires gentle air thinning to prevent pooling.
Adhesive layer thickness
Excessive pooling of adhesive should be avoided, as overly thick adhesive layers (several hundred microns) may act as a weak mechanical interface and appear radiolucent on radiographs.17 Conversely, an adequately thick adhesive layer can function as a stress-absorbing intermediate layer during composite polymerization. A flexible adhesive layer may accommodate polymerization shrinkage stresses by slight elastic deformation.18 Additionally, because the outermost portion of resin (around 10 to 20 µm)19 is subject to oxygen inhibition, a thin adhesive layer may experience incomplete polymerization. Studies have shown improved bond strength when certain thin adhesives are applied in two layers (with intermediate curing)20 or covered with the adhesive (second bottle) of a two-bottle adhesive or a flowable composite.21,22
Light polymerization of the adhesive layer
Adequate light energy is required for complete adhesive polymerization. Energy delivery depends on both curing light intensity and exposure time. Research within the author’s lab has demonstrated that curing an adhesive for three seconds with high-intensity light may achieve similar bond strength to curing for 20 seconds at lower intensity, whereas a one-second exposure is insufficient even at higher power. Proper positioning of the curing light is also essential to ensure adequate energy delivery to the adhesive layer. It is also preferrable to place and polymerize overlaying composite soon after adhesive polymerization to prevent time for osmosis of water through the hybrid layer from underlying dentin.23
Prevention of hydrolytic and enzymatic degradation of the hybrid layer
Long-term durability of dentin bonding remains a challenge due to enzymatic and hydrolytic degradation. Enzymatic degradation results from matrix metalloproteinases (MMPs) present within dentin, which are activated during acid etching and degrade exposed collagen fibrils. Application of 2% chlorhexidine following etching has been shown to inhibit MMP activity.24 Hydrolytic degradation occurs when water penetrates the hybrid layer from dentinal tubules and causes hydrolysis of chemical groups, particularly in adhesives with high hydrophilic content. Therefore, minimizing voids, ensuring complete resin infiltration, and limiting excessive hydrophilicity within the hybrid layer are essential for improving long-term bond durability.
Editor's note: This article appeared in the April 2026 print edition of Dental Economics magazine. Dentists in North America are eligible for a complimentary print subscription. Sign up here.
References
- Van Meerbeek B, Yoshihara K, Van Landuyt K, Yoshida Y, Peumans M. From Buonocore's pioneering acid-etch technique to self-adhering restoratives: a status perspective of rapidly advancing dental adhesive technology. J Adhes Dent. 2020;22(1):7-34. doi:10.3290/j.jad.a43994
- Perdigão J. Current perspectives on dental adhesion: (1) dentin adhesion—Not there yet. Jpn Dent Sci Rev. 2020;56(1):190-207. doi:10.1016/j.jdsr.2020.08.004
- Oliveira SS, Pugach MK, Hilton JF, Watanabe LG, Marshall SJ, Marshall GW Jr. The influence of the dentin smear layer on adhesion: a self-etching primer vs a total-etch system. Dent Mater. 2003;19(8):758-767. doi:10.1016/S0109-5641(03)00023-X
- Saikaew P, Sattabanasuk V, Harnirattisai C, Almas Chowdhury AFM, Carvalho R, Sano H. Role of the smear layer in adhesive dentistry and the clinical applications to improve bonding performance. Jpn Dent Sci Rev. 2022;58:59-66. doi:10.1016/j.jdsr.2021.12.001
- Almeida G, Marques JA, Van Meerbeek B, Ramos JC, Falacho RI. Particle abrasion as a pre-bonding dentin surface treatment: a scoping review. J Esthet Restor Dent. 2024;36(5):746-760. doi:10.1111/jerd.13183
- Dhanpal P, Yiu CKY, King NM, Tay FR, Hiraishi N. Effect of temperature on water sorption and solubility of dental adhesive resins. J Dent. 2009;37(2):122-132. doi:10.1016/j.jdent.2008.10.004
- Delgado AHS, Ahmed MH, Nunes Ferreira M, Mano Azul A, Polido M, Yoshihara K, Van Meerbeek B. Physico-chemical properties and performance of functional monomers used in contemporary dental adhesive technology. J Adhes Dent. 2025;27:175-193. doi:10.3290/j.jad.c_2297
- De Munck J, Mine A, Poitevin A, Van Ende A, Cardoso MV, Van Landuyt KL, Peumans M, Van Meerbeek B. Meta-analytical review of parameters involved in dentin bonding. J Dent Res. 2012;91(4):351-357. doi:10.1177/0022034511431251
- de Paris Matos T, Perdigão J, de Paula E, Coppla F, Hass V, Scheffer RF, Reis A, Loguercio AD. Five-year clinical evaluation of a universal adhesive: a randomized double-blind trial. Dent Mater. 2020;36(11):1474-1485. doi:10.1016/j.dental.2020.08.007
- Hashimoto M, Ohno H, Endo K, Kaga M, Sano H, Oguchi H. The effect of hybrid layer thickness on bond strength: demineralized dentin zone of the hybrid layer. Dent Mater. 2000;16(6):406-411. doi:10.1016/S0109-5641(00)00035-X
- Tsujimoto A, Shimatani Y, Nojiri K, Barkmeier WW, Markham MD, Takamizawa T, Latta MA, Miyazaki M. Influence of surface wetness on bonding effectiveness of universal adhesives in etch-and-rinse mode. Eur J Oral Sci. 2019;127(2):162-169. doi:10.1111/eos.12596
- Nonato RF, Moreira PHA, Silva DOD, Ferreira MWC, Reis A, Cardenas AFM, Loguercio AD, Siqueira FSF. Long-term evaluation of bonding performance of universal adhesives based on different dentinal moisture levels. J Adhes Dent. 2022;24:395-406. doi:10.3290/j.jad.b3559027
- Carvalho GLM, Moreira PM, Carneiro BT, Suzuki TYU, Lanza MDS, Alvim HH, Giannini M, Andre CB. Impact of active vs passive application of dental adhesives on bond strength to dentin and enamel: a systematic review and meta-analysis of in vitro studies. Jpn Dent Sci Rev. 2025;61:90-102. doi:10.1016/j.jdsr.2025.04.001
- Loguercio AD, Raffo J, Bassani F, Balestrini H, Santo D, do Amaral RC, Reis A. Twenty-four-month clinical evaluation in noncarious cervical lesions of a two-step etch-and-rinse adhesive applied using a rubbing motion. Clin Oral Investig. 2011;15(4):589-596. doi:10.1007/s00784-010-0408-8
- Luque-Martinez IV, Perdigão J, Muñoz MA, Sezinando A, Reis A, Loguercio AD. Effects of solvent evaporation time on immediate adhesive properties of universal adhesives to dentin. Dent Mater. 2014;30(10):1126-1135. doi:10.1016/j.dental.2014.07.002
- Tang C, Mercelis B, Yoshihara K, Peumans M, Van Meerbeek B. Does the universal adhesive's film thickness affect dentin-bonding effectiveness? Clin Oral Investig. 2024;28(2):150. doi:10.1007/s00784-024-05523-7
- Van Meerbeek B, Willems G, Celis JP, Roos JR, Braem M, Lambrechts P, Vanherle G. Assessment by nano-indentation of the hardness and elasticity of the resin-dentin bonding area. J Dent Res. 1993;72(10):1434-1442. doi:10.1177/00220345930720101401
- Shawkat ES, Shortall AC, Addison O, Palin WM. Oxygen inhibition and incremental layer bond strengths of resin composites. Dent Mater. 2009;25(11):1338-1346. doi:10.1016/j.dental.2009.06.003
- Ahmed MH, Yao C, Van Landuyt K, Peumans M, Van Meerbeek B. Extra bonding layer compensates universal adhesive's thin film thickness. J Adhes Dent. 2020;22(5):483-501. doi:10.3290/j.jad.a45179
- Fujiwara S, Takamizawa T, Barkmeier WW, Tsujimoto A, Imai A, Watanabe H, Erickson RL, Latta MA, Nakatsuka T, Miyazaki M. Effect of double-layer application on bond quality of adhesive systems. J Mech Behav Biomed Mater. 2018;77:501-509. doi:10.1016/j.jmbbm.2017.10.008
- Montes MA, de Goes MF, da Cunha MR, Soares AB. A morphological and tensile bond strength evaluation of an unfilled adhesive with low-viscosity composites and a filled adhesive in one and two coats. J Dent. 2001;29(6):435-441. doi:10.1016/S0300-5712(01)00037-9
- Tay FR, Pashley DH, Suh BI, Carvalho RM, Itthagarun A. Single-step adhesives are permeable membranes. J Dent. 2002;30(7-8):371-382. doi:10.1016/S0300-5712(02)00064-7
- Montagner AF, Sarkis-Onofre R, Pereira-Cenci T, Cenci MS. MMP inhibitors on dentin stability: a systematic review and meta-analysis. J Dent Res. 2014;93(8):733-743. doi:10.1177/0022034514538046
About the Author
Nathaniel Lawson, DMD, PhD
Dr. Lawson is an associate professor and the director of the division of biomaterials at the University of Alabama at Birmingham School of Dentistry. He graduated from UAB School of Dentistry in 2011 and obtained his PhD in biomedical engineering in 2012. He has lectured internationally, served as an investigator on numerous research grants, and published extensively on dental materials. He also works as a general dentist in the UAB Faculty Practice.
Updated September 25, 2023
Ting-Chia Liu, BDS, MDS
Dr. Liu is a general dentist from Taiwan currently pursuing a master of science and residency in dental biomaterials at the University of Alabama at Birmingham. Her work focuses on operative and esthetic dentistry, prosthodontic applications of contemporary dental materials, and digital technologies in modern dental care.
Pouya Sabanik, DDS, PgC
Dr. Sabanik is a resident and master of science student in dental biomaterials at the University of Alabama at Birmingham School of Dentistry. He completed a postgraduate certificate in restorative dentistry at the University of Michigan School of Dentistry. Having earned his DDS from Tabriz University of Medical Sciences, his research focuses on digital dentistry, dental ceramics, and biomaterials.