Blinded by my sight: have to see it to defeat it!
by Patti DiGangi, RDH, BS
I recently went to the restroom of a nice restaurant and stood in the line that’s so often experienced by women. Two stalls for such a big restaurant were just not sufficient. I waited patiently while a mother with three young children attempted to take care of her personal needs - no easy task. The kids, being kids, punched, jumped, and played in the sink. When one of the children ran behind me, I turned to see what would be the next mischief. Lo and behold, there was a stall behind me that I had not seen when I entered the rest- room. My brain clouded my ability to see; I was blinded by my sight. How we see what we see is not only a function of our eyes, but also our brains. Improving vision means increasing what our eyes see and stretching our brains to interpret what is exposed.
This often happens with routine soft tissue oral cancer screenings; we make assumptions based on previous experiences, or we’re distracted and don’t see what’s there. Other times we just can’t see. Oral cancer is a disfiguring affliction that kills half its victims. In the past, it has been difficult to detect in its early stages, but this is no longer true. Using early detection options in addition to standard measures, oral health professionals can change this, yet we seem to resist adopting new options.
Heart disease and oral cancer
Early in 2007, public broadcasting (www.pbs.org) aired a special called “The Hidden Epidemic - Heart Disease in America.”1 There are many parallels and connections with the early detection of heart disease and the early detection of oral cancer. One in two men and one in three women in the U.S. will develop heart disease during their lifetimes.2 For many, the first sign is a massive heart attack. Since the 1950s, the angiogram had been the gold standard for detecting blocked arteries. Over six million operations or other interventions for heart disease are performed in American hospitals each year.1 Many of them are a patient’s second, third, or fourth procedure.
Cardiologists often thought there had to be something more going on, something they couldn’t see. A new technology, the Intravascular Ultrasound (IVUS), a miniaturized ultrasound probe, scans the cells that line the artery walls. IVUS is an adjunct to coronary angiography and coronary angioplasty. While angiography continues to be the gold standard for coronary imaging, it is becoming increasingly important to understand the anatomy within the artery wall. IVUS facilitates a more accurate measurement of the degree of stenosis of the opening of the coronary arteries than angiography. Dr. Peter Libby, chief of Cardiovascular Medicine at the Brigham and Women’s Hospital in Boston, said it well: “If you want to defeat a disease, you’ve got to see it.”1
Long before there is overt disease breakdown, there are changes. The majority of oral cancers are still not being caught until Stage III or IV, with more than 50 percent of victims dead in five years. These statistics have not significantly improved for 40 years. In the past three decades, a 60 percent increase in tongue cancer has been detected in people less than 40.3-6 This information, along with the 25 percent of oral cancer cases with no known risk factors, caused researchers to look for new causes. Human papillomavirus (HPV) is known to cause cervical cancer, but until now, its connection with oral cancer was based primarily on molecular data. An epidemiologic study published in May 2007 in the New England Journal of Medicine has now provided compelling support for this relationship.7 The statistics on HPV are staggering, with approximately 20 million people currently infected.8-10 The most dangerous HPVs, 16 and 18, are transmitted through sexual contact.10 In 2006, the Centers for Disease Control (CDC) voted to recommend first vaccine Gardasil®, developed to prevent cervical cancer and other diseases caused by certain types of HPVs.11 The vaccine unfortunately does not treat existing HPV infections, genital warts, precancers, or cancers, making risk assessment and early detection even more important.
Just as with heart disease, something more is needed to detect early changes that lead to oral cancer. Oral health research has brought new early detection modalities to market. Most professionals have heard about early detection systems, yet only about 5 percent have adopted them for routine use. It’s been said everything is new in dentistry for 10 to 20 years. This may have been true and acceptable in the past, but this is the 21st century and it is time for more professionals to update their thinking and methods.
Early detection options for immediate action
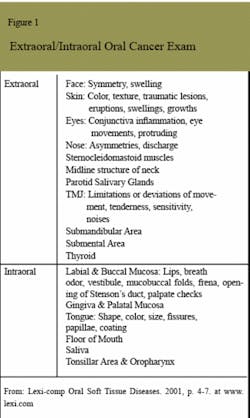
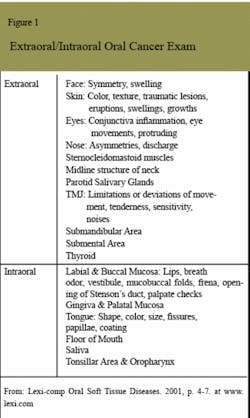
The gold standard for oral cancer detection remains the extraoral/intraoral soft tissue exam (see Figure 1). The first step is risk assessment, which includes a thorough review of the health profile. The highest risk factors remain tobacco and alcohol, yet many other factors need to be assessed. Even using good risk assessment, the fact remains that 25 percent of oral cancer victims have no lifestyle risk factors, and this group is the most rapidly growing patient segment.
Changes occur when plaque accumulates in the arteries and leads to heart disease. This cannot be felt or detected with some of the most common tests. Changes can occur and lead to oral cancer even when we can’t feel or detect them with our routine systems. What Dr. Libby said about heart disease is also true about oral cancer - if we want to defeat it, we have to see and detect those early changes.
Current options
In dentistry, there are several different options for improving visualization. Early detection modalities are not meant to replace standard screening but improve its yield.
1) Brush biopsy: Dental professionals often see what appear to be harmless looking, unexplained red and white spots. Oral brush biopsy is a noninvasive method of evaluating oral mucosal lesions for cellular atypia. It is a three-layer transepithelial exfoliative cytology technique. Computerized brush biopsy analysis became commercially available in 1999. The Oral CDx® (CDx Laboratories, Inc. Suffern, N.Y., www.oralcdx.com) is an easy and painless test for those unexplained spots that may be precancer or cancer. Each testing kit costs between $32 and $38.
2) Chemiluminescence technology: Even before there are any visual or tactile clues, change is occurring on the cellular level. Rinsing with a dilute acetic acid solution helps visualization of abnormal squamous epithelium tissue that appears acetowhite when viewed under diffuse low-energy wavelength light. Normal epithelium will absorb the light and appear dark. The enhanced ViziLite Plus system (Zila Pharmaceuticals, Inc., Phoenix, Ariz., www.vizilite.com) was introduced in October 2005.12 This system combines the oral screening using chemiluminescent light technology with TBlue630, a marking system using Zila Tolonium Chloride. Zila’s Tolonium Chloride (ZTC™) is a patented composition designed to be superior to traditional toluidine blue. The cost of one ViziLite examination kit ranges from $19.95 to $29.95 per unit.
3) Fluorescence technology: Due to the limitations of white light examination of the oral mucosa, it is possible for clinicians to identify tissue changes on and below the surface before they become apparent under white light examination. VELscope® (LED Dental Inc., White Rock, British Columbia, www.velscope.com), which was developed in partnership with the British Columbia Cancer Agency, was approved for use in the U.S. in April 2006. VELscope is a hand-held device that emits a safe blue light into the oral cavity. This excites the tissue from the surface of the epithelium to the basement membrane and into the stroma beneath, causing it to fluoresce. The clinician is then able to view the different fluorescence responses to differentiate between normal and abnormal tissue.
As reported in the July issue of DE, VELscope received clearance from the FDA in April 2007 for two expanded indications: “VELscope is intended to be used by a dentist or health-care provider as an adjunct to traditional oral examination by incandescent light to enhance the visualization of oral mucosal abnormalities that may not be apparent or visible to the naked eye, such as oral cancer or premalignant dysplasia. VELscope is further intended to be used by a surgeon to help identify diseased tissue around a clinically apparent lesion and thus aid in determining the appropriate margin for surgical excision.” The retail price for the VELscope is $4,995, with approximately $5 per exam in disposables cost.
Exciting future
The future continues to unfold. Researchers at the School of Dentistry at UCLA laboratories have discovered possible biomarkers in saliva for the detection of oral cancer. Cancer researchers have traditionally used blood serum and urine to detect cancer signatures. Saliva contains the same biomarkers as those found in blood. Nanotechnology enabled scientists to uncover the components of saliva, including the distinct messenger ribonucleic acids (RNAs) that serve as oral cancer signatures, on an atomic or molecular scale. The Saliva RNA Test analyzes for the presence of four statistically significant oral cancer biomarkers and is undergoing clinical testing.12-17
There may soon be another FDA-approved option called OraTest®, which is a patented five-minute mouth rinse that uses a stabilized and patented formulation of toluidine blue. OraTest is already approved in more than a dozen countries.18-19
These are exciting times with so many new developments and options pending.
Oral Cancer Testing and CDT 2007-2008 Codes
Insurance codes and interpretations are constantly changing in our rapidly shifting, quickly evolving world. The newest version of the dental benefit coding book is now time-stamped, as shown in its name - CDT 2007-2008. This newest version is time-sensitive and will become obsolete Jan. 1, 2009. Practices and practitioners need to be aware of the new, changed, and deleted codes by utilizing the most current version.
The clinical oral evaluation section underwent several important changes. Codes D0150, Comprehensive Oral Evaluation; D0120, Periodic Oral Evaluation; and D0180, Comprehensive Oral Evaluation have been altered to include specific language on oral cancer evaluation. The Code Revision Committee report stated the reason for this change: “Expansion of language to include oral cancer evaluation based on growing awareness of change needs to reduce morbidity and mortality of oral cancer.” Because this language is specifically part of these codes, to submit these codes without performing oral cancer evaluation could constitute fraud. The only way to demonstrate that the evaluation was performed is to complete the appropriate documentation.
The latest information and guidance for using CDT codes and medical codes for the brush biopsy, chemiluminescence, and fluorescence adjunctive-testing technologies are available on the Web sites of each manufacturer.
Don’t hesitate any longer
A patient may have no symptoms or warning, and using traditional tools, dentists may be misled into thinking that someone is fine, when in fact, hidden trouble may be brewing. The development and routine use of early detection modalities sets the stage for a fundamental change in the early detection of oral cancer. Dental health professionals can help reduce the morbidity and mortality of oral cancer. We have better ways to see it, which creates ways to defeat it.
One of the strongest assets of human beings is the ability to form habits. A downside to those same habits is that we can take the path of least resistance and resist change. As professionals, we want to do what is best. Yet, even when we know something better is available, the desired behavior does not spontaneously occur. We can get into a bit of a rut, especially if we stay in the same routine. We may be able to temporarily change our behavior, but then autopilot kicks in and we fall back into old behaviors, even when they don’t serve us very well.
We don’t have to wait and shouldn’t hesitate when excellent adjunctive options are available now. With our increasing ability to reduce the horrendous morality rates for oral cancer, it is unfortunate that adoption of new technologies is relatively slow and inconsistent. As Mahatma Gandhi said, “We must become the change we want to see.”
References
1. The Hidden Epidemic - Heart Disease in America, 2007 WGBH Educational Foundation. Available at: http://www.pbs.org/wgbh/takeonestep/index.html. Accessed Feb. 14, 2007.
2. National Center for Statistics: Heart Disease, Center for Disease Control. January 24, 2007. Available at: http://www.cdc.gov/nchs/fastats/heart.htm Accessed Feb. 27, 2007.
3. Cancer prevention, early detection, and prediction. National Cancer Institute. Available at: http://www.cancer.gov/aboutnci/2015/cancer-Prevention-Early-Detection-and-Prediction. Accessed March 9, 2006.
4. Rates of occurrence in the United States. February 2006. The Oral Cancer Foundation Web site. Available at: http://www.oralcancerfoundation.org/facts/. Accessed April 13, 2005.
5. Head and neck cancer. November 2005. Oncology Channel Web site. Available at: http://www.oncologychannel.com/headneck/index.shtml. Accessed July 26, 2005.
6. What are the risk factors for oral cavity and oropharyngeal cancer? March 2006. American Cancer Society Web site. Available at: http://www.cancer.org/docroot/CRI/content/CRI_2_4_2X_What_are_the_risk_factors_for_oral_cavity_and_oropharyngeal_can cer_60.asp?sitearea=CRI. Accessed July 26, 2005.
7. D’Souza G, Kreimer AR, Viscidi R, et al. “Case-control study of human papillomavirus and oropharyngeal cancer.” N Engl J Med. 2007 May 10;356(19):1944-1956. Available at: http://content.nejm.org/cgi/content/short/356/19/1944. Accessed Aug. 20, 2007.
8. Human Papillomaviruses and Cancer: Questions and Answers. June 8, 2006. National Cancer Institute. Available at: http://www.cancer.gov/cancertopics/factsheet/Risk/HPV. Accessed June 17, 2006.
9. Waldman H. Vaccine Treatment Takes Aim At Oral Cancer. June 6, 2006. UConn Health Center. Available at: http://cancer.uchc.edu/news/releases/oralcancer.html. Accessed June 17, 2006.
10. Fakhry C, Gillison M. Clinical Implications of Human Papillomavirus in Head and Neck Cancers, J Clin Oncol 2006 24: 2606-2611 Available at: www.oral cancerfoundation.org/facts/humanpapillomavirus.htm. Accessed Aug. 2, 2006.
11. HPV Vaccine Questions and Answers. June 2006. Centers for Disease Control. Available at: http://www.cdc.gov/std/HPV/STDFact-HPV-vaccine.htm. Accessed Aug. 2, 2006.
12. Wong D. “Salivary diagnostics powered by nanotechnologies, proteomics and genomics” J Am Dent Assoc 2006 137: 313-321. Available at: http://jada.ada.org/cgi/content/abstract/137/3/313. Accessed April 25, 2006.
13. Shagat S. “UCLA School of Dentistry researchers have standardized a saliva-based test for oral cancer, ushering in an era of new possibilities for cancer detection.” UCLA News. March 9, 2006. Available at: http://news room.ucla.edu/page.asp?RelNum=6883. Accessed April 25, 2006.
14. Malamud D. “Salivary diagnostics: The future is now.” J Am Dent Assoc 2006 137: 284-286.
15. Zila to Launch ViziLite® Plus with TBlue630™ at Annual ADA Meeting. Business Wire; 10/5/2005. Available at: www.highbeam.com/library/docFree.asp?DOCID=1G1:137135123. Accessed July 31, 2006.
16. Bringing the promise of molecular medicine to oral cancer screening. Inside Scoop, National Institute of Dental and Craniofacial Research. March 2006. Available at: www.nidcr.nih.gov/NewsAndReports/Media/InsideScoop3162007.htm. Accessed July 31, 2006.
17. Ozols J. Healthy Mouths. Newsweek. Dec. 22, 2005. Available at: http://msnbc.msn.com/id/6746583/site/newsweek/print/1/displaymode/1098/. Accessed June 17, 2006.
18. Zila Begins Phase III OraTest® Clinical Trial with Investigators’ Meeting. Healthcare Sales and Marketing Network. December 13, 2005. Available at: http://salesandmarketingnetwork.com/news_release.php?ID=2009008. Accessed June 17, 2006.
19. New oral test detects genetic alterations predictive of oral and head and neck cancers. 2001. Abramson Cancer Center University of Pennsylvania. Available at: http://www.oncolink.com/resources/article.cfm?c=3&s=8&ss=23&id=1472&month=07&year=2001 Accessed June 17, 2006.
Patti DiGangi, RDH, BS, is a speaker, author, and practicing dental hygiene clinician. Her passions are early oral cancer detection, eagle-eye assessments, minimal intervention, oral health/whole body wellness, and dental insurance. She has been speaking nationally since 1993 and brings a wealth of experience, knowledge, and spontaneity to her presentations. DiGangi can be reached through her Web site at www.pdigangi.com.