ORAL CANCER: A CHALLENGE TODAY, A CONQUEST TOMORROW
by Kevin D. Huff, DDS, MAGD
Oral cancer mortality rates and the incidence of oral cancers have remained relatively unchanged for decades. Today, if you are diagnosed with late-stage oral cancer — which is when most oral cancer is discovered — and are treated appropriately, you have about a 55% to 60% chance of living for five years. Someone dies of oral or oropharyngeal cancer every hour of every day. It is our responsibility as health-care professionals to continue to challenge the status quo of oral mucosal screening technologies. Our patients’ lives depend on it!
However, the demand and criteria for evidence-based standards in dentistry coupled with public expectations of the role of dentists in health care create a challenging environment for innovative thinkers. For example, I personally have scared patients away from my care by having broached the topic of HPV and sexual history prematurely and possibly even inappropriately. By these mistakes, I have learned some finesse and restraint in my approach to taking patient histories. The general public seems to be resistant to the idea of dentists becoming involved in detailed medical history reviews that may include details of personal sexual history, and many dentists feel that exploring medical histories in-depth beyond the obvious relationship to dental health is outside the scope of general dentistry — or at least outside of their comfort zones.
This bimodal challenge creates inherent conflict between morality and the principles of dental ethics. Nearly every dentist whom I have asked over the past several years went into dentistry to help others. So, if it is accepted that the status quo of oral cancer survival rates is unacceptable, then it stands to reason that generally dentists want to do what they can to make a difference. The question What should I do? is one of morality.
Fig. 1 — Two independent studies in a general dental practice setting yield similar conclusions about direct tissue fluorescence visualization using the VELscope. The Huff study was a retrospective study conducted in a single low-risk general dental practice. The Truelove study was a prospective, controlled study with multiple examiners.
Probably the greatest challenge to the general implementation of adjunctive oral mucosal screening devices is the reported lack of high-level research studies that are double-blinded, multicentered, and reproducible. Most of the research available is related to case reports or extrapolation of literature from gynecological medicine, which is the origin of the concept of using light after dehydration with acetic acid for oral mucosal screening. To put those who question the lack of research at ease, I am happy to report that now at least two peer-reviewed studies exist that support the efficacy of direct tissue fluorescence visualization, beyond the initial research publications required for FDA 501K clearance, using specifically the VELscope system (LED Dental, Inc., Burnaby, BC, Canada) (Fig. 1).
However, social pressures and arguments about the lack of research, along with the ambiguity of how to proceed if some type of adjunctive screening system suggests disease, make it tough to decide whether dentists should “jump on the bandwagon” with adjunctive screening devices or sit idly by on the sidelines doing what we’ve always done (conventional oral cancer exams). In other words, the dentist is forced to ask Why should I do it? It is a question of ethics. If we continue to do what we have been doing, will we make enough of a difference in oral cancer mortality and morbidity? Could it be that we simply are not doing enough? There really is no proverbial fence for the dentist to ride when it comes to oral cancer screening. If we continue to do what we have done, we will continue to get what we have always gotten; if we add adjunctive screening systems to our protocol, then we are truly sailing on uncharted waters.
Oral cancer is a real burden to society. According to SEER data, the lifetime risk of developing oral or oropharyngeal cancer is higher than other well-known killer cancers such as lymphoma and brain cancer. Oral cancer affects one out of 152 women and one out of 71 men, and there were an estimated 39,400 new cases of oral cancer discovered in 2011, with about 640,000 new cases discovered worldwide each year. At this rate, with the current survival rate at approximately 60%, only about 19,000 of those Americans diagnosed today will be alive in 2016!
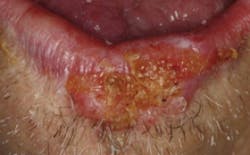
Mature oral cancer lesions show classic signs that can be easily recognized during a properly performed conventional oral exam. Typically, oral cancer presents as firm, indurated lesions that exhibit surface irregularity and lack of mucosal mobility. They often show variation of color, and they may present as slowly healing or nonhealing ulcerations. The lesions usually progressively increase in size if left untreated, and there is usually altered sensation from the lesion. Sometimes these lesions are painful; sometimes they just may be numb. Although there is a guideline that a lesion dimension of more than 10 millimeters is of concern, a hallmark of oral squamous cell carcinoma is a distinct margin. Based on these signs and symptoms, one may easily argue that oral cancer can and should be detected with conventional examination techniques, which is the argument made by naysayers of adjunctive screening techniques (Fig. 2).
Fig. 2 — Squamous cell carcinoma of the lip. The lesion has distinct borders, variegation of color, induration, and ulceration. There is a complete loss of sensation of the lower lip, and the lip does not move during speech.
Early lesions may present with much more subtlety. Dysplastic precancerous lesions appear red, white, or mixed white and red — erythroplakia, leukoplakia, or erythroleukoplakia. The problem is that an early variant of red is pink, virtually indistinguishable from normal oral epithelium with the naked eye. Here are some key points to remember about epithelial dysplasia:
- 70% of red lesions will become cancer within five years.
- 25% of white lesions will become cancer within five years.
- Once progression to cancer occurs, most of those lesions metastasize rapidly.
There is a distinction between a five-year survival rate of a primary cancerous lesion and subsequent chances for other primary tumors. This is the concept of field cancerization, which is likely due to embryological development patterns. Because of this unique presentation pattern of head and neck cancer, it must be understood that if a person is diagnosed with a cancerous lesion and even survives five years without recurrence, that individual has a very high risk of producing more primary tumors in his or her lifetime — up to 35 times more risk than if they had not had a first lesion! Often, death is due to the second primary tumor. This may be due to a lack of understanding about field cancerization and/or poor compliance on the patients’ part regarding follow-up evaluations for a lifetime, because they think that they have passed the hallmark five years.
Once a patient has been diagnosed with oral cancer, careful follow-up oral cancer screening exams at least yearly are critical. In my opinion, those patients who have been diagnosed with precancerous lesions should be followed with similar diligence as well. These are high-risk patients!
It may be surprising that many dentists are fearful of managing patients who have been previously treated for oral cancer. For example, two years ago, a 68-year-old nonsmoker who had been treated for oral squamous cell carcinoma 10 years prior presented to my office as a new patient. Before he sat down in my chair, this otherwise healthy and friendly gentleman paused to give me a firm handshake, thanking me with tears in his eyes for my willingness to see him for a dental problem that he was having. As he revealed his story to me, it became apparent that this fellow had contacted 10 general dental offices over the past several years to become established for regular maintenance and hygiene recare. Because of his history, he mentioned during every initial phone call to each practice that he was an oral cancer survivor. All 10 dental offices told him that they “didn’t treat oral cancer cases.”
Fig. 3 — Recurrent squamous cell carcinoma of the soft palate.
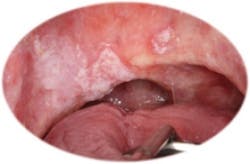
During his exam in my office, I found a substantial lesion (Fig. 3) extending across the midline — a mixed, firm lesion with irregular borders, which was raised above the surrounding tissues. There was generalized loss of fluorescence around the hyperkeratotic areas of the lesion that did not blanch under pressure (Fig. 4). Referral for biopsy to an ENT physician was made immediately, and the patient ended up losing his entire soft palate and part of his hard palate due to recurrent squamous cell carcinoma. It was impossible to determine what that lesion may have looked like or what the outcome of the treatment for this secondary cancerous lesion would have looked like had one of those 10 other dentists seen him several years before, recognized that something appeared abnormal, and referred for appropriate diagnosis and management.
Fig. 4 — Image of the lesion in Fig. 3 taken through the VELscope Vx. Loss of fluorescence indicates dysplastic activity, and the increased fluorescence represents increased keratinization.
Aside from a previous history of oral cancer, there are many other factors that play a role in the potential for a person to develop oral cancer. Traditionally, age and gender have been considered risk factors. It has already been mentioned that men tend to be more at risk than women. In the absence of comorbid factors, men over the age of 50 tend to have the highest risk, especially if they happen to be black. So, ethnicity is also a factor. The largest risk factors seem to be the combined use of tobacco and alcohol, accounting for as much as 75% of oral squamous cell carcinomas that present anterior to the tonsillar pillars. The discovery and studies related to the involvement of the human papillomavirus — most commonly Type 16 — show a rising incidence of oropharyngeal cancer in young men who do not use tobacco and alcohol, as well as a growing presentation in young women. Some researchers also feel that other chronic conditions, such as candidiasis, lichen planus, and the herpes simplex virus, as well as poor oral hygiene practices and periodontal disease, may play a part in increasing an individual’s risk of developing oral cancer.
During the past decade, it has been interesting — albeit challenging — to be a part of the race to develop “the magic bullet” against oral cancer. Although management of diagnosed oral cancer has undergone its own transformation in medical research, I still believe that the best chance we have in improving oral cancer survival rates is through detection of early lesions — the “what-is-its” that have the potential to become cancer. I am convinced that the state-of-the-art of oral cancer screening needs to be continually improved. Therefore, I am an advocate for any development that encourages or helps the clinician perform an enhanced examination.
While reflective visual technologies using acetic acid washes are still in practice, they are limited by the individual costs for each patient, patient tolerance to the wash, and the fact that they are best suited for enhancing the visualization of leukoplakias and are not very effective for identifying much more concerning subtle erythroplakias. The addition of a proprietary solution of toluidine blue to better visualize and photograph suspicious lesions has improved its use. However, I believe that there are more promising adjunctive visualization devices available today.
Direct tissue fluorescence technologies use light in the visible light spectrum to provide information about the condition of the oral mucosa. One proposed mechanism of action is the photo-excitation of Krebs cycle enzymes in epithelial cells that biofluoresce. A secondary method of action is the fluorescence of intact collagen cross-links in the stroma of healthy epithelium. While several systems are available that use direct tissue fluorescence visualization, the VELscope system was the first of its kind and remains, in my opinion, the premier adjunctive visual screening system on the market — still owned by its original manufacturer and developer, LED Dental, Inc., which is now a publicly traded company.
The most important factor for me in determining which direct tissue fluorescence visualization system to choose is the ease of ability to photograph what is seen. A key component to the decision about whether or not a biopsy is needed is the persistence of the lesion for more than two weeks. Unless a photograph is taken, one has difficulty assessing change. Furthermore, photographing a lesion under fluorescence is needed for communication with specialists to whom a patient is referred, because the fluorescent appearance is substantially different from the clinical appearance under white light. Currently, the VELscope Vx is the only system on the market that has this capability. In fact, an easy-to-use camera is also available as part of the Vx system.
While a surgical biopsy remains the gold standard for obtaining a true diagnosis of a suspicious clinical lesion, there have been improvements in the cytology systems available. For example, the OralCDx BrushTest® (Suffern, N.Y.) improved protocol now involves sending in the entire collection brush in a liquid medium for review in addition to the cytological smear prepared chairside. This minimizes the chance of missed cells that would otherwise have been discarded on the brush. OralAdvanceTM (Vancouver, BC, Canada) is a liquid cytology system that has been developed to qualitatively assess chromosomal levels in individual cells, called ploidy, which suggests oncogenic potential. A few university pathology departments continue to offer liquid cytology analyses, similar to cervical PAP smears.
Salivary diagnostics continue to move forward, but the implementation of these systems remains a challenge for the general practitioner. For example, there are no clear protocols for what a clinician should do with a patient who tests positive for HPV on a saliva test without clinical symptoms. There is also a challenge with the cost vs. the benefit of the test. However, I am very excited by the interest from the manufacturers of OraRisk® (Oral DNA Labs, Brentwood, Tenn.) to move forward in search of a better way and in their progressive search for how to collaborate with other adjunctive screening system manufacturers in the fight against oral cancer.
With the growth of laser implementation in dentistry, surgical biopsy technique trends are also changing. While the scalpel biopsy technique is best to preserve pristine margins, many clinicians are using lasers to perform adequate biopsies. The risk of laser biopsy techniques is charring of potentially involved margins. However, the most exciting innovation in surgical biopsy techniques is the use of direct tissue fluorescence visualization to determine the surgical margins, which is one of the FDA-approved indications for the VELscope.
The future of adjunctive mucosal screening is bright, and research continues. From my experience “in the trenches” of oral mucosal screening and on the lecture circuit speaking with many amazing clinicians, the utopia of adjunctive screening would look like this:
- All laboratories reviewing cytology or oral tissue samples would employ board-certified oral pathologists for rendering valid assessments and diagnoses.
- The cost of adjunctive screening systems would be at a price point that all dentists could have multiple units and be able to provide screening exams at a minimal cost to the patient.
- All adjunctive visualization systems would lend themselves easily to photography.
- Rather than individualized saliva tests for specific entities, a general salivary assay — similar to a medical blood assay — would be available at minimal cost to the dentist that would qualitatively and quantitatively provide general information about bacterial strains and disease entities in the saliva that could then be used for clinical treatment planning, decision-making, and subsequent outcomes assessment of treatment.
- Quality continuing education courses reviewing oral cancer screening technology, basic pathology, and enabling general dentists to perform surgical biopsies appropriately should continue to be offered regularly in an ethical fashion.
In conclusion, morality is what we know to be inherently right or inherently wrong — it is a spiritual concept. Ethics is a legalistic approach to reasoning, which may be in conflict with an individual clinician’s morality. One may argue that adjunctive oral cancer devices have weak supporting evidence, may lead to overtreatment, may create unnecessary expenses, etc. However, the same person may also believe that adjunctive oral cancer screening technologies help provide better patient care and that implementing them is the right thing to do. These two statements are diametrically opposed, creating a dilemma. For me, the implementation of adjunctive oral cancer screening technologies into my practice is both moral and ethical, and I am thankful to be able to offer them as a service to my patients, because the current survival rates of oral cancer are unacceptable to me.
Kevin D. Huff, DDS, MAGD, is a practicing general dentist in Dover, Ohio, and lectures in the United States and Canada on oral cancer early detection. He is a clinical instructor at the Case School of Dental Medicine, and the coordinator of oral mucosal screening at the Mercy Medical Center GPR Program in Canton, Ohio. You may contact him by email at [email protected].
Disclosure:
Dr. Huff has no financial interests to disclose and has received no compensation from any company for authoring this article.
HPV
The human papillomavirus, especially Type 16, transforms epithelial cells in the genital and upper respiratory tract by invading the chromosomal DNA to cause oncogenic mutations. These mutations produce tumor suppression proteins through transcription to allow uncontrolled proliferation of infected mutated cells. Oncoproteins are also produced that stimulate cellular proliferation and shut down cell cycle regulation and controlled cell death (apoptosis). The subsequent uncontrolled cellular proliferation results in a lack of cellular differentiation and ongoing mutations from mitosis of unstable chromosomes.
The current understanding is that HPV has been primarily implicated in squamous cell carcinoma of the oropharynx, which is posterior to the anterior tonsillar pillars. In fact, most tonsillar cancers are known to be infected with HPV-16. HPV-16 has also been identified in refractory periodontal pockets as well, and at least one study has shown a correlation with periodontal pocket depth and tongue cancer.
HPV does not appear to be associated with epithelial dysplasia. However, cervical lymphadenopathy is typically present in HPV-related cancers. Unilateral tonsillar induration firmness should raise a concern for the examiner. Currently, however, there is no consensus about the appearance of a clinical precursor to an HPV tumor.
Past DE Issues